MANAGING BEST PRACTICES: Making sense of the zeros
For example, how are you going to describe OSHA’s new permissible exposure limit (PEL) and action level for hexavalent chromium (assuming these levels have not been changed from the proposed standard)?

Comparisons
If the PEL and/or action level appear in a list with other exposure limits, keeping all the units the same may help the reader draw relative comparisons, as in example Table I. Examples of comparison lists, keeping the units in mg/m3, appear in OSHA’s Table Z-1 at 29 CFR 1910.1000 and in the ACGIH® TLV® booklet.
Visual and verbal confusion?
As we increase the numbers to the right of the decimal place, however, visual confusion may be introduced. At a glance, was the action level for hex chrome three or four zeros after the decimal place?
Also, the action level for hex chrome is awkward to say as it appears. We usually wouldn’t want to say that the action level is “point zero, zero, zero five milligrams per cubic meter.â€
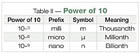
Power of 10
When we move zeros, and other numbers, to the right of the decimal place, the concept of the power of 10, raised to a negative number, is introduced. Table II summarizes this concept.Table II is used most often when we verbally describe or write an exposure limit or measurement, usually by itself. For example, the PEL for total dust may be described as “fifteen milligrams per cubic meter†or written 15 mg/m3. And generally, we’d say that the PEL for carbon black is “three point five milligrams per cubic meter.â€
However, when there are two or more places to the right of the decimal point, people generally start using the micro prefix. The lead PEL may be described as “fifty micrograms per cubic meter.†And hex chrome would be described as “one microgram per cubic meter.â€
If convention holds, then the action level for hex chrome would generally be stated as “point five micrograms per cubic meter†or “one-half microgram per cubic meter.†But it may be just as proper to say that the action level for hex chrome is “500 nanograms per cubic meter.â€
Mixing the prefixes together may be confusing and should be avoided when describing groups of chemicals. If you need to explain the PEL for several chemicals at the same time, try to keep their units, i.e. prefix, the same.
Prefix significance
A jump from the micro to the nano prefix is extremely significant, but is often taken too casually. A micro is one-millionth of something. A nano is one-billionth of something. How do the two compare? Look at it this way, in a time comparison one part per million (micro) is equal to about 30 seconds in one year. One part per billion (nano) is equal to about one second in 32 years! The time concept hits home when you really think about it, and not just read it. We should stress what the prefix is when we say it to someone. Don’t casually jump from one prefix to another.Nano world
We’re entering into a nano world, no doubt about it. Look at the proposed Threshold Limit Value (TLV®) for beryllium at 0.0002 mg/m3. How do you want to describe it? If we describe it as a whole number, it will have the nano prefix.The Lumex field instrument for measuring mercury vapor can detect concentrations as low as two nanograms per cubic meter. So what’s the big deal? It can detect mercury vapors in your mouth if you have certain types of dental fillings.
Technology is allowing us to measure, and report, things in extremely small concentrations. And in our risk-adverse society, we often find that the only exposure concentration that will make some people happy is an all-zero exposure. It doesn’t seem to matter if you have a lot of zeros to the right of the decimal point, as soon as you place any real number (i.e. 1, 2, 3, etc.), you may have a problem. Many people don’t want a nano-exposure — they want no exposure.
“Less thanâ€
Another problem you may encounter is we may never know how small an exposure really was or is. All laboratories (that I am aware of) will report non-detect analytical measurements using the less than “<†symbol. There will always be doubt if some, however small, concentration of the substance may have been present.No easy answer
There is no easy answer to how you explain all this to someone else. First, you need to judge their level of understanding. Have they had math and science classes before? Do they understand the “powers of 10†that are raised to a negative number? Do they know and appreciate the real meaning behind prefixes such as “micro†and “nano?†And most importantly, what’s their belief on exposure and risk?Risk communication experts such as Peter Sandman caution us in using concentration comparisons (e.g., one part per billion is equal to one second in 32 years) when explaining risk, or we may “damage our credibility.†We must be careful when we try to explain risks.
As technology moves forward, one thing is certain: we’re likely to see more zeros describing exposures and measurements. From my experience, you’ll need patience when trying to explain this to someone else.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




